Which Family of Elements Contains a Full Outer Shell of Electrons?
Learning Outcomes
- Define the octet rule and its office in chemical bonds
Not all elements have enough electrons to fill their outermost shells, but an atom is at its almost stable when all of the electron positions in the outermost shell are filled. Considering of these vacancies in the outermost shells, we encounter the formation of chemical bonds, or interactions between two or more than of the aforementioned or different elements that result in the formation of molecules. To attain greater stability, atoms will tend to completely fill their outer shells and will bond with other elements to attain this goal past sharing electrons, accepting electrons from some other cantlet, or altruistic electrons to some other atom. Because the outermost shells of the elements with low atomic numbers (up to calcium, with atomic number xx) tin hold eight electrons, this is referred to as the octet dominion. An element tin can donate, accept, or share electrons with other elements to fill its outer shell and satisfy the octet rule.
An early on model of the atom was developed in 1913 by the Danish scientist Niels Bohr (1885–1962). The Bohr model shows the cantlet as a central nucleus containing protons and neutrons, with the electrons in circular electron shells at specific distances from the nucleus, like to planets orbiting effectually the sun. Each electron shell has a unlike energy level, with those shells closest to the nucleus beingness lower in free energy than those further from the nucleus. By convention, each shell is assigned a number and the symbol n—for example, the electron vanquish closest to the nucleus is chosen 1n. In order to move between shells, an electron must absorb or release an corporeality of energy corresponding exactly to the difference in energy between the shells. For example, if an electron absorbs energy from a photon, it may go excited and move to a higher-energy shell; conversely, when an excited electron drops back down to a lower-energy trounce, it will release energy, often in the form of oestrus.
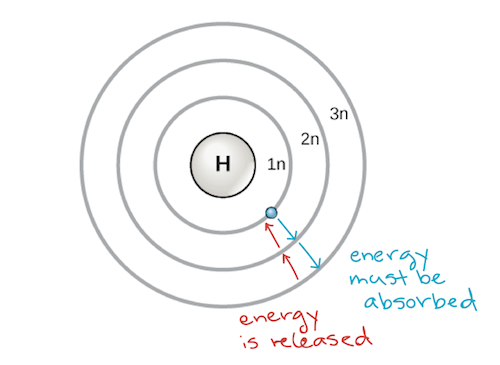
Bohr model of an atom, showing energy levels as concentric circles surrounding the nucleus. Free energy must be added to move an electron outward to a higher energy level, and energy is released when an electron falls downward from a college free energy level to a closer-in one. Paradigm credit: modified from OpenStax Biology
Atoms, like other things governed by the laws of physics, tend to have on the everyman-energy, most stable configuration they can. Thus, the electron shells of an atom are populated from the inside out, with electrons filling upward the low-free energy shells closer to the nucleus before they motion into the college-free energy shells farther out. The shell closest to the nucleus, 1n, tin hold two electrons, while the next shell, 2n, can hold eight, and the third shell, 3n, can concord up to eighteen.
The number of electrons in the outermost beat out of a particular atom determines its reactivity, or tendency to grade chemical bonds with other atoms. This outermost shell is known equally thevalence crush, and the electrons found in it are calledvalence electrons. In general, atoms are most stable, least reactive, when their outermost electron shell is full. Most of the elements important in biology need 8 electrons in their outermost vanquish in order to be stable, and this rule of pollex is known equally theoctet rule. Some atoms tin can be stable with an octet even though their valence shell is the 3n shell, which tin can agree up to xviii electrons. We will explore the reason for this when nosotros talk over electron orbitals beneath.
Examples of some neutral atoms and their electron configurations are shown beneath. In this table, you tin come across that helium has a full valence shell, with two electrons in its offset and only, 1n, trounce. Similarly, neon has a complete outer 2n beat containing eight electrons. These electron configurations make helium and neon very stable. Although argon does non technically have a full outer shell, since the 3n shell tin can hold up to eighteen electrons, information technology is stable like neon and helium because it has eight electrons in the 3n shell and thus satisfies the octet rule. In dissimilarity, chlorine has only seven electrons in its outermost shell, while sodium has but one. These patterns do not fill the outermost shell or satisfy the octet rule, making chlorine and sodium reactive, eager to gain or lose electrons to reach a more stable configuration.
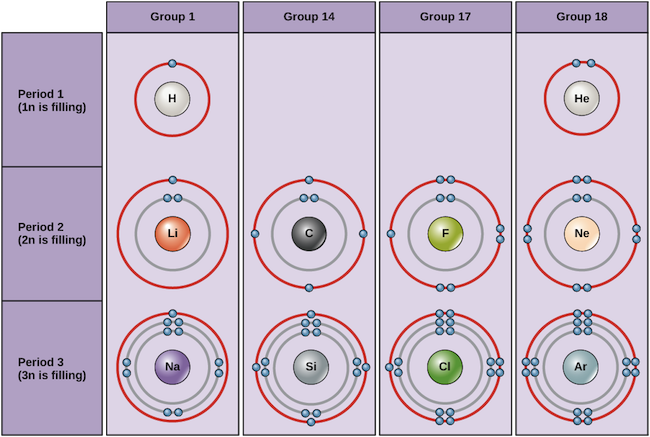
Bohr diagrams indicate how many electrons fill each chief shell. Group 18 elements (helium, neon, and argon) accept a full outer, or valence, trounce. A full valence shell is the nearly stable electron configuration. Elements in other groups take partially filled valence shells and gain or lose electrons to achieve a stable electron configuration.
Electron configurations and the periodic table
Elements are placed in gild on the periodic table based on their atomic number, how many protons they accept. In a neutral cantlet, the number of electrons will equal the number of protons, so we can easily determine electron number from atomic number. In add-on, the position of an element in the periodic table—its column, or group, and row, or period—provides useful information about how those electrons are arranged.
If we consider just the first iii rows of the table, which include the major elements important to life, each row corresponds to the filling of a different electron beat out: helium and hydrogen identify their electrons in the 1n shell, while second-row elements similar Li start filling the 2n trounce, and 3rd-row elements like Na continue with the 3n shell. Similarly, an element's column number gives information about its number of valence electrons and reactivity. In full general, the number of valence electrons is the aforementioned within a column and increases from left to right inside a row. Group ane elements have merely 1 valence electron and group 18 elements have eight, except for helium, which has only two electrons total. Thus, group number is a good predictor of how reactive each chemical element volition exist:
- Helium (He), neon (Ne), and argon (Ar), as group 18 elements, accept outer electron shells that are full or satisfy the octet rule. This makes them highly stable as unmarried atoms. Because of their not-reactivity, they are called theinert gases ornoble gases.
- Hydrogen (H), lithium (Li), and sodium (Na), every bit group 1 elements, have only ane electron in their outermost shells. They are unstable equally single atoms, but can become stable by losing or sharing their one valence electron. If these elements fully lose an electron—every bit Li and Na typically exercise—they become positively charged ions: Li+, Na+.
- Fluorine (F) and chlorine (Cl), as grouping 17 elements, have seven electrons in their outermost shells. They tend to achieve a stable octet by taking an electron from other atoms, becoming negatively charged ions: F− and Cl−.
- Carbon (C), as a group xiv element, has four electrons in its outer shell. Carbon typically shares electrons to achieve a complete valence shell, forming bonds with multiple other atoms.
Thus, the columns of the periodic table reflect the number of electrons found in each element's valence beat out, which in plough determines how the element volition react.
Effort It
Contribute!
Did you have an idea for improving this content? We'd dear your input.
Ameliorate this pageLearn More
strongallontention.blogspot.com
Source: https://courses.lumenlearning.com/wm-nmbiology1/chapter/chemical-bonding/
0 Response to "Which Family of Elements Contains a Full Outer Shell of Electrons?"
Post a Comment